Acute Lymphoblastic Leukemia
On this page
Overview
What is Acute lymphoblastic leukemia (ALL)?
Acute lymphoblastic leukemia (ALL), also called acute lymphocytic leukemia, is a type of cancer that occurs in white blood cells called lymphocytes. These cells play a role in the body's immune response to illness and disease. Lymphocytes develop in the bone marrow and are found in the blood and all parts of the lymphatic system, including the lymph nodes, thymus and spleen.
ALL accounts for 20 percent of acute leukemia in adults but is the most common type of acute leukemia in children. If left untreated, this form of cancer typically gets worse quickly.
Our approach to acute lymphoblastic leukemia
UCSF is dedicated to delivering the most advanced treatment options for acute lymphoblastic leukemia (ALL) with care and compassion. Our goal is to achieve a cure. Standard treatments are effective in curing ALL in most adults, but for certain high-risk cases, we offer additional therapies, including stem cell transplants. Allogeneic transplantation – which uses stem cells or bone marrow from a tissue-matched donor – is our first choice, but if a compatible donor isn't available, we offer autologous transplantation, which uses cells collected from the patient.
Risk factors for ALL
In most cases, experts can't determine what causes someone to develop acute lymphocytic leukemia (ALL). However, certain factors may increase your risk of developing ALL. These factors include:
- Past treatment with chemotherapy or radiation therapy
- Exposure to high levels of radiation
- Long-term exposure to toxic chemicals like benzene
- Certain genetic disorders, including Down syndrome, Fanconi anemia and neurofibromatosis
Symptoms of ALL
Acute lymphoblastic leukemia (ALL) usually causes illness suddenly, within days or weeks. Most symptoms are related to the reduction of healthy bone marrow and a decrease in normal blood counts. ALL causes bone marrow failure, which leads to:
- Fatigue and shortness of breath, due to low levels of red blood cells
- Easy bleeding and bruising, due to low platelet counts
- Many infections, due to low counts of normal white blood cells
In addition to these symptoms, ALL can sometimes cause enlarged lymph nodes, liver or spleen, as well as pain in the bones or joints.
Diagnosis of ALL
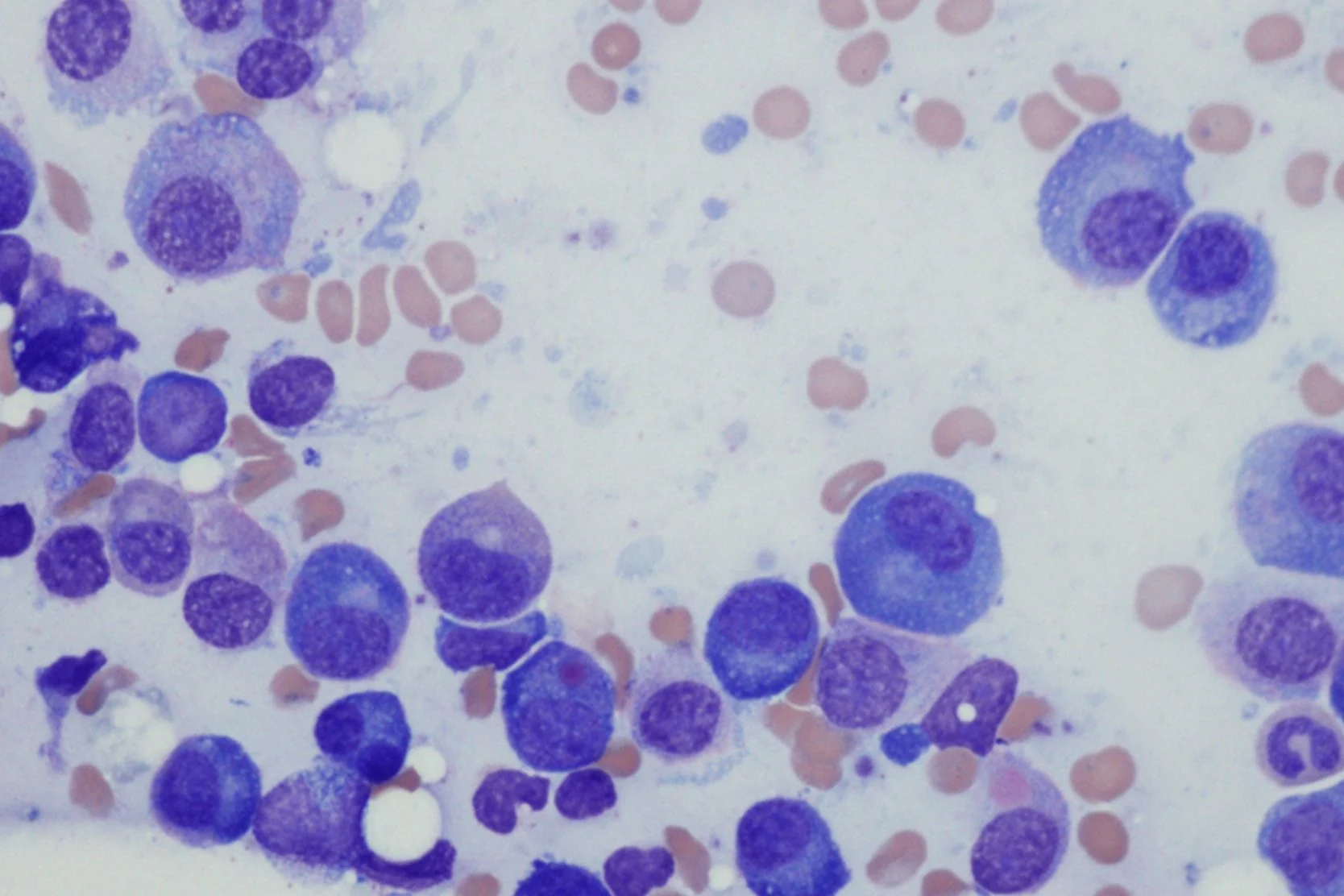
Acute lymphoblastic leukemia (ALL) is usually suspected when a blood test finds abnormal blood counts and leukemic cells, or blasts, appear in the blood. It's generally difficult to be certain of an ALL diagnosis simply by the appearance of cells under the microscope. Therefore, additional laboratory tests are normally needed. These tests include:
- Bone marrow aspiration and biopsy. ALL is diagnosed if the bone marrow contains 20% or more immature cells called blasts, determined to be lymphoid in nature.
- Immunophenotyping. Also called flow cytometry, this evaluation of the type of proteins expressed by the cells determines whether they are lymphoid (ALL) or myeloid (which would indicate a different cancer, acute myeloid leukemia, or AML). It also determines whether the affected white blood cells are T or B lymphocytes.
- Cytogenetics. A type of chromosome testing that is a critical part of the evaluation that helps determine the best course of treatment.
Treatment of ALL
The treatment of acute lymphoblastic leukemia (ALL) varies depending on one's age, general condition at diagnosis and the results of the cytogenetic testing. Typically, chemotherapy is the primary treatment for ALL. However, depending on the subtype, other therapies may be considered. These can include drug therapy, immunotherapy, or a stem cell or bone marrow transplant. Some patients may also be eligible to participate in investigative treatments.
Chemotherapy treatment can be divided into these four phases:
- First phase: Induction chemotherapy
- Second phase: Consolidation chemotherapy
- Third phase: Maintenance chemotherapy
- Fourth phase: Central nervous system (CNS) prophylaxis
The entire treatment process for ALL typically continues for two to three years.
Complete remission – a cure – is achieved in about 90% of patients, with 25 to 40% enjoying long-term survival. About 5% of patients die of treatment-related complications during their initial therapy, and another 5% never go into remission.
Chemotherapy for ALL
Treatment of ALL is usually urgent and needs to begin within days, and sometimes the same day, as the diagnosis is made. The first phase of treatment, called induction chemotherapy, requires that patients remain in the hospital for approximately four weeks.
Induction chemotherapy
The most common induction chemotherapy drugs used for ALL are daunorubicin, vincristine, prednisone, asparaginase and sometimes cyclophosphamide.
During this phase, patients receive intensive supportive care, including transfusion of red blood cells and platelets. Antibiotics and anti-fungal medications are used to treat and prevent both bacterial and fungal infections. A medication known as G-CSF (Neupogen) can be used to help quickly reestablish a normal white blood count.
In rare cases, patients may develop mouth sores and issues with their intestinal tract. Complete but temporary hair loss does occur.
Once blood counts have returned to normal, another bone marrow biopsy is performed to determine if the patient has entered complete remission. A complete remission is achieved when the blood and bone marrow show no evidence of persistent leukemia and blood counts have returned to normal.
Consolidation chemotherapy
Chemotherapy drugs used during consolidation include the same ones used during induction, as well as Ara-C, etoposide, methotrexate and 6-mercaptopurine.
The consolidation phase typically includes multiple cycles of intensive chemotherapy given over a six- to nine-month period. Frequent hospitalizations and intensive supportive care are still needed, including red blood cell and platelet transfusions.
Maintenance chemotherapy
Once patients have completed intensive chemotherapy, they need to take oral chemotherapy pills for an additional 18 to 24 months. These pills – typically methotrexate and 6-mercaptopurine – are usually well-tolerated with only minimal side effects.
Patients need to have blood tests once a month while taking chemotherapy pills. Most patients with ALL can return to work during maintenance therapy.
Central nervous system (CNS) prophylaxis
ALL often recurs in the cerebrospinal fluid – the fluid that bathes the spinal column and brain. To prevent this, chemotherapy is injected directly into the cerebrospinal fluid. This is done by inserting a needle between the vertebrae of the lower back – called a spinal tap or lumbar puncture – and infusing chemotherapy directly into the clear cerebrospinal fluid. This procedure is called intrathecal chemotherapy.
Patients routinely have six rounds or more of intrathecal chemotherapy to prevent recurrence of ALL. More injections may be necessary if leukemia cells are detected in the cerebrospinal fluid.
Most people complete intrathecal therapy within two to four months of starting their treatment. Headaches and nausea are occasional side effects.
Supportive drug therapies for ALL
ALL patients are prone to viral infections and to a pneumonia called pneumocystis during their treatment. Antibiotics, such as acyclovir and Septra or Bactrim, are given during ALL therapy to help prevent these infections.
Up to one-quarter of adults with ALL have the Philadelphia chromosome in their leukemia cells, which often contributes to a poor prognosis. There are several FDA-approved drugs available to these patients, including imatinib (Gleevec) and dasatinib (Sprycel). Patients begin taking these drugs during induction chemotherapy and continue indefinitely. A stem cell transplant is typically the best treatment option for ALL patients with the Philadelphia chromosome.
Stem cell transplant for ALL
Stem cell transplantation, also called blood or marrow transplantation (BMT), is only performed in patients who have abnormal cytogenetics (chromosome testing) or other features of high-risk ALL. Cytogenetics is the most important factor in deciding whether a person should have a bone marrow transplant for ALL. Patients with the Philadelphia chromosome or with the translocation involving chromosomes 4 and 11, should go on to BMT.
At UCSF Medical Center, the preferred standard therapy is allogeneic transplantation which uses stem cells or bone marrow from a matched donor, ideally a sibling. Young patients with very high-risk ALL who need an allogeneic transplant, but lack a matched sibling donor, are sometimes matched to an unrelated donor though the National Marrow Donor Program (NMDP).
For patients with high-risk ALL who lack compatible donors, UCSF offers autologous transplantation, which uses cells collected from the patient's own blood after they have achieved complete remission. Patients who are having an autologous transplant first undergo radiation therapy and chemotherapy.
Then the stem cells are collected from the blood using a technique called apheresis.
In the apheresis procedure, an IV catheter is inserted into one of the large veins in the neck. This catheter is connected to an apheresis machine, which separates the blood cells, allowing the collection of only the white blood cells, which are then frozen All the other cells, including red blood cells and platelets, are returned to the patient.
Each apheresis procedure takes four hours. Two to three apheresis procedures are usually necessary to collect enough stem cells. After completing radiation and chemotherapy, the collected stem cells are thawed out and re-infused through the vein to restart blood production.
The hospitalization for the high-dose radiation, chemotherapy and re-infusion of autologous peripheral blood stem cells lasts approximately four weeks. The side effects are severe and aggressive supportive care measures are needed.
Immunotherapy for ALL
Immunotherapy works by helping your body's immune system fight off cancer cells. Instead of attacking the cancer directly, this treatment boosts your body's natural defense systems. Immunotherapy may be an option for people whose leukemia has come back after other treatments, or for those who aren't able to have a stem cell transplant.
Investigational therapies for ALL
UCSF is dedicated to improving outcomes of ALL patients through studies of promising treatments, known as clinical trials. Interested patients may have opportunities to participate in clinical trials for ALL treatments. Learn more about the process and benefits of participating in a clinical trial.
Awards & recognition

Best in California and No. 7 in the nation for cancer care
220+
blood and marrow transplants performed each year
Recommended reading
Related services & treatments
Specialties
Treatments
- Allogeneic Stem Cell Transplant
- Allogeneic Bone Marrow Transplant
- Autologous Stem Cell Transplant
- Stem Cell Transplant
- Bone Marrow Transplant
Helpful resources
Support services
UCSF Health medical specialists have reviewed this information. It is for educational purposes only and is not intended to replace the advice of your doctor or other health care provider. We encourage you to discuss any questions or concerns you may have with your provider.