Acute Myeloid Leukemia
On this page
Overview
What is acute myeloid leukemia (AML)?
Acute myeloid leukemia (AML), also called acute myelogenous leukemia, is a cancer of the blood and bone marrow. It occurs when your bone marrow produces large numbers of abnormal white blood cells. AML is the most common type of acute (rapidly developing and progressing) leukemia in adults, accounting for 80% of cases. Without treatment, it usually gets worse quickly.
Our approach to acute myeloid leukemia
UCSF is dedicated to delivering the most advanced treatment options for AML with care and compassion. All patients begin with an extensive evaluation, which includes a chromosomal examination called cytogenetic testing. This analysis can reveal information that helps us determine the best course of treatment. Patients typically receive two rounds of chemotherapy and, in some cases, a stem cell transplant.
Our highly skilled team provides intensive supportive care to address side effects and protect patients from infection and other risks that can accompany treatment. Our providers' expertise also includes stem cell transplantation, (also known as blood and marrow transplantation), through the internationally renowned UCSF Hematology, Blood and Marrow Transplant, and Cellular Therapy (HBC) Program.
We are dedicated to discovering better treatments for AML through research. Interested patients may have opportunities to participate in clinical trials for acute myeloid leukemia treatment. Learn more about the process and benefits of participating in a clinical trial.
Symptoms of acute myeloid leukemia
Typically, symptoms of acute myeloid leukemia (AML) come on suddenly – over days or weeks. Less often, a patient is ill for a few months or has a history of one of the bone marrow disorders categorized as a myelodysplastic syndrome.
AML makes you sick by interfering with your normal bone marrow function. Instead of producing healthy white blood cells, the bone marrow starts making abnormal ones that divide and multiply uncontrollably. These abnormal cells replace and crowd out the normal cells in your bloodstream, leading to low numbers of red blood cells, white blood cells and platelets.
When you don't have enough red blood cells, you can develop anemia, a condition that causes fatigue. When your platelet counts are low, you can become prone to bleeding and bruising, especially in the skin, nose and gums.
A low white blood cell count puts you at an increased risk of infection. The infection types vary, but signs of a possible infection include:
- Fever
- Runny nose
- Cough
- Chest pain
- Shortness of breath
- Pain when urinating
- Occasional diarrhea
If your white blood cell count gets too low, your ability to fight off infection may be seriously compromised. This makes you vulnerable to developing potentially life-threatening conditions, such as pneumonia (a lung infection) or sepsis (a severe response to infection).
Diagnosis of acute myeloid leukemia
Diagnosing acute myeloid leukemia (AML) usually involves the following:
- Physical examination. The doctor will check for swelling of your liver and spleen and for swollen lymph nodes in your groin, neck and armpits.
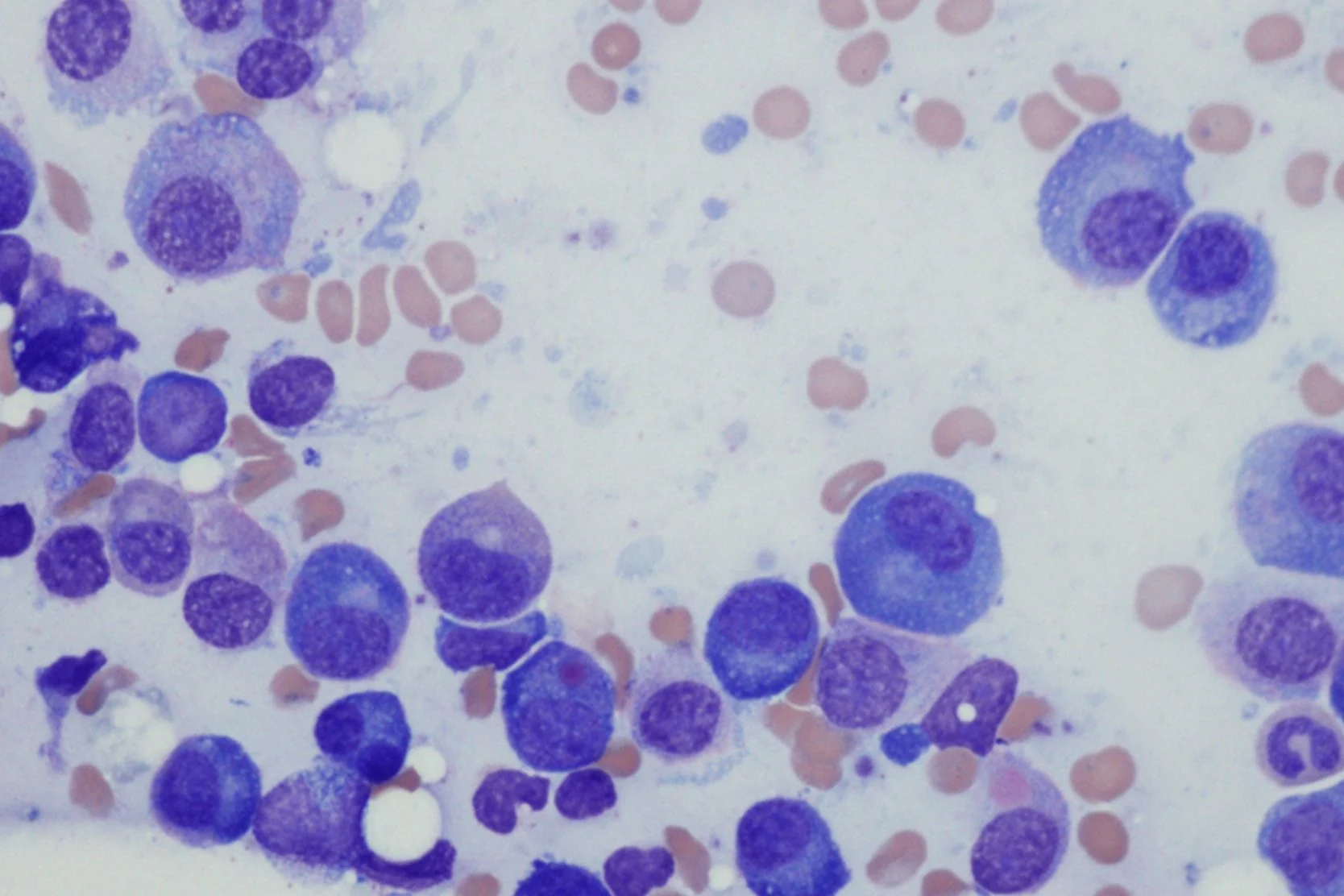
- Blood tests. The laboratory will analyze a sample of your blood in various ways, such as measuring the amounts of red blood cells, white blood cells and platelets. A pathologist will also examine your blood cells under a microscope to see whether they show abnormalities.
- Bone marrow aspiration and biopsy. These procedures are often done together to check for leukemia cells and to identify the type of AML you have. The doctor removes a small amount of liquid bone marrow and a piece of the spongy bone marrow tissue, which are then examined under a microscope.
AML is diagnosed when at least 20% of the white blood cells in your bone marrow are immature cells called blasts. Normally, less than 5% of the cells in bone marrow are blasts.
Additional tests are performed to confirm the diagnosis, to determine the type of AML and to assess how aggressive it's likely to be. Immunophenotyping, a method of analyzing the proteins expressed by cells, can help identify the subtype. In addition, a small amount of fluid bone marrow is sent for cytogenetic testing, which looks for changes in chromosomes. Information from this test can be useful in making treatment decisions and predicting long-term outlook.
Types of acute myeloid leukemia
There are two major types of AML. The distinction has to do with how different the leukemia cells are from normal blood cells and the specific genetic abnormalities. The types progress differently and require different treatments.
The two major types are:
- Acute myeloid leukemia, non-APL. Overproduction of immature cells impacts the production of healthy white and red blood cells and platelets. The outlook for non-APL AML depends on whether there are changes to the chromosomes of the leukemia cells.
- Acute promyelocytic leukemia (APL). APL makes up 10 to 15% of AML cases. Symptoms and complications of APL generally involve bleeding, bruising and blood clots. Thanks to the development of APL-specific therapies, the chances of a successful treatment are good.
Treatment of acute myeloid leukemia
Because AML can progress rapidly, it's important to start treatment as soon as possible. The specific treatment plan depends on the patient's age, overall health and results of their cytogenetic testing. For most patients under 70, the treatment goal is to cure their AML.
Typically, AML therapy is performed in two phases:
- First phase. Induction chemotherapy uses drugs to destroy as many cancer cells as possible.
- Second phase. Called consolidation chemotherapy, this part of treatment aims to destroy any remaining cancer cells, helping prevent a recurrence. The second phase may include stem cell transplantation.
First phase: Induction chemotherapy for AML
Induction chemotherapy uses anticancer drugs to destroy as many cancer cells as possible. The goal is remission, which is when no leukemia cells are detectable in the blood or bone marrow and the body maintains normal numbers of blood cells without the need for transfusions.
You'll be hospitalized for this treatment phase, as the chemotherapy drugs have serious side effects. Your white blood cell count will drop low, making you susceptible to infections. Your red blood cell count will also go down, resulting in fatigue, weakness and other symptoms of anemia.
Our skilled and experienced team will monitor and care for you throughout this process. They may give you transfusions of red blood cells and platelets to correct anemia and prevent bleeding. You'll also receive medications to prevent bacterial and fungal infections or to treat any that occur. A drug called filgrastim (Neupogen) may also be used to help prevent infections by restoring your white blood cell count more quickly.
Side effects of induction chemotherapy can include temporary hair loss, sores in the mouth, and throat and skin rashes. You may receive drugs to prevent or reduce nausea and diarrhea, eye drops to prevent irritation and fortified beverages to improve your nutrition.
Most patients are in the hospital for four to five weeks. You'll be discharged once your blood counts have returned to normal and your bowels are functioning properly. At the end of your induction therapy, you'll have a bone marrow biopsy to see whether your AML is in remission. Approximately 70 to 80% of patients enter complete remission after this first phase of treatment.
Those who achieve complete remission are given a one-month break to prepare for the second phase of treatment.
Second phase: Consolidation chemotherapy for AML
This treatment phase aims to destroy any remaining abnormal cells that may not be active but could cause a recurrence of the cancer. Treatment entails additional cycles of intensive chemotherapy and, in some cases, a bone marrow transplant (BMT), also known as a stem cell transplant.
Consolidation chemotherapy uses medications similar to those used in the first treatment phase, but the number of chemo cycles in phase two varies from case to case.
In general, side effects of consolidation treatment are similar to those of induction therapy, requiring intensive support. Most patients stay in the hospital during the four to five weeks of treatment, though some institutions provide consolidation chemotherapy on an outpatient basis. Overall, approximately 30 to 40% of patients receiving consolidation chemotherapy are cured of their AML.
High-dose chemotherapy and blood or bone marrow transplant (BMT) for AML
Some patients with aggressive AML require a bone marrow transplant. There are two types:
- Autologous stem cell transplantation. This uses the patient's own stem cells. Before the procedure, stem cells are harvested from the patient and then the patient receives extra high doses of chemotherapy to destroy any residual cancer cells. The treatment has serious side effects, and going through it is difficult. But about half of the AML patients who complete the therapy are cured.
- Allogeneic stem cell transplantation. This uses stem cells from a matched donor, who may be a close relative or a stranger. (Matching means that certain proteins on the cells' surfaces are compatible.) The patient receives high-dose chemotherapy to destroy residual cancer cells before receiving the stem cells. An allogeneic transplant can cure the disease but is typically used only in the most serious cases because it's more difficult and dangerous than an autologous transplant or chemotherapy alone.
Treatment for acute promyelocytic leukemia (APL)
The treatment plan is different for patients who have the type of AML called acute promyelocytic leukemia (APL). Therapy for APL includes a vitamin A derivative called all-trans retinoic acid (ATRA) and arsenic trioxide, together with standard chemotherapy drugs. Most people undergo multiple cycles of this therapy. APL patients rarely need a stem cell transplant. Because of advances in diagnosis and treatment, APL is now considered the most curable form of acute myeloid leukemia.
Awards & recognition

Best in California and No. 7 in the nation for cancer care

Rated high-performing hospital for leukemia, lymphoma & myeloma care
Recommended reading
Related services & treatments
Specialties
Treatments
- Cancer Chemotherapy
- Blood and Bone Marrow Transplant
- Stem Cell Transplant
Helpful resources
Support services
UCSF Health medical specialists have reviewed this information. It is for educational purposes only and is not intended to replace the advice of your doctor or other health care provider. We encourage you to discuss any questions or concerns you may have with your provider.