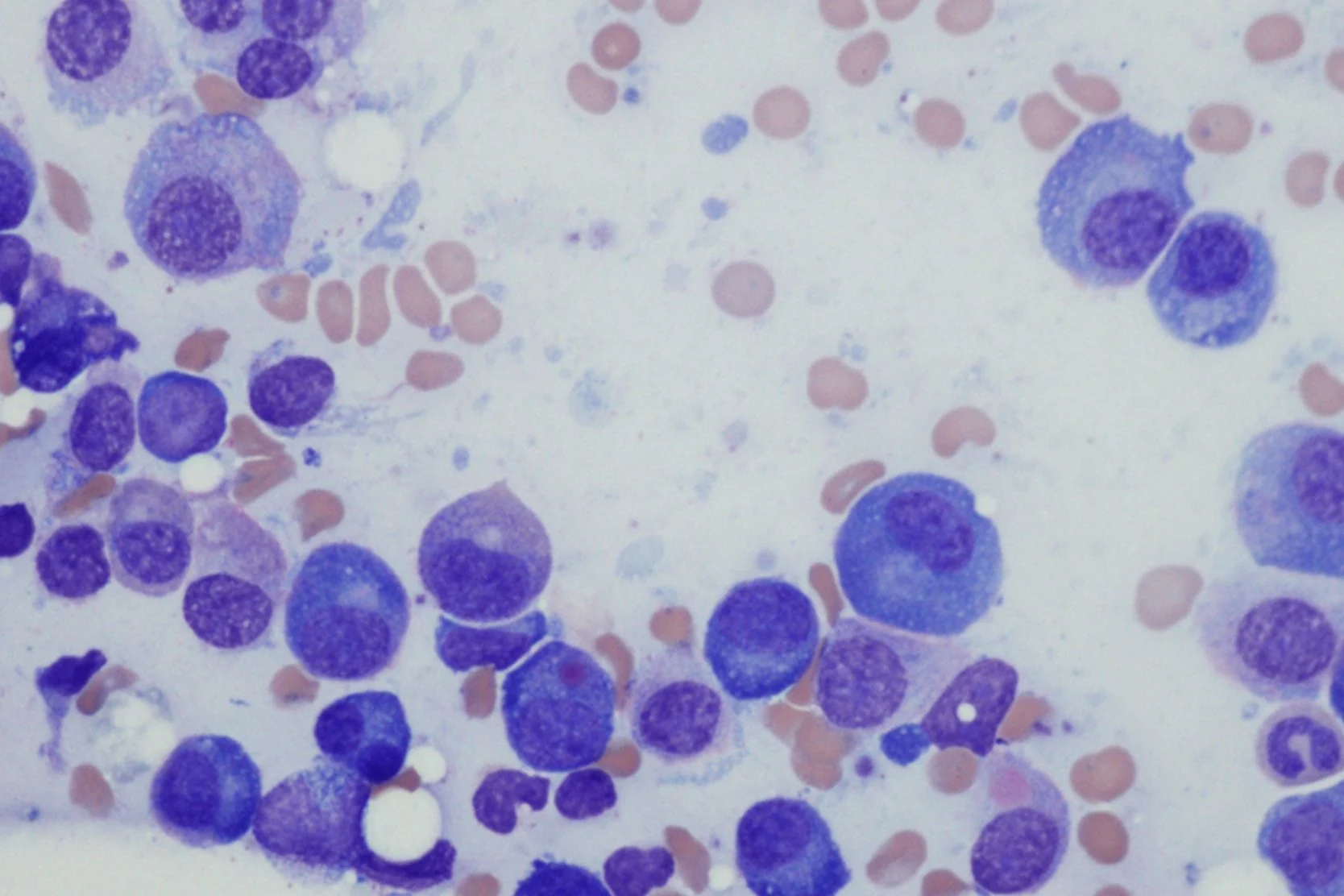
Chronic Lymphocytic Leukemia
On this page
Overview
Chronic lymphocytic leukemia (CLL) is a disorder in which the lymphocytes lose their normal ability to die and end up accumulating over time. At first, the cells increase only in the blood, but over years they also increase in the lymph nodes, liver, spleen and bone marrow.
Some patients with CLL live decades and die of infection or other diseases common in older adults.
Our approach to chronic lymphocytic leukemia
UCSF delivers the most advanced treatment options for chronic lymphocytic leukemia (CLL) with care and compassion. Most patients with early-stage CLL do not need treatment. As the condition advances, we have a number of chemotherapy drugs that will put it into remission for several years. In select cases, where the potential benefits outweigh the risks, we offer stem cell transplantation using tissue-matched donors.
We are also dedicated to discovering better treatments for CLL through research. Interested patients may have the option to participate in clinical trials of potential new therapies.
Signs & symptoms
Many patients have no symptoms at the time of diagnosis, except for an elevated white blood cell count. Some patients notice fatigue, enlargement of lymph nodes or fullness in the abdomen due to an enlarged spleen.
When CLL becomes more advanced and begins to replace normal bone marrow, low blood counts can cause:
- Anemia, due to low levels of red blood cells
- Infections, due to low levels of white blood cells called neutrophils
- Bleeding, due to low levels of platelets
CLL also increases a person's risk of infection because of low production of antibodies (gamma globulins) that help fight bacteria.
In 5 percent to 10 percent of cases, CLL causes the destruction of the patient's own red blood cells and/or platelets through an "autoimmune" process. Destruction of platelets is called immune thrombocytopenic purpura (ITP), and destruction of red blood cells is called autoimmune hemolytic anemia (AHA).
Diagnosis
Chronic lymphocytic leukemia is diagnosed through the detection of a high white blood cell count composed primarily of small lymphocytes. The diagnosis is confirmed by "immunophenotyping" (flow cytometry), which determines the types of proteins on the leukemia cells. CLL classically expresses CD19 (a B-lymphocyte protein) and CD 5 (a T-cell lymphocyte protein) simultaneously.
CLL is the one type of leukemia for which there is a formal staging system:
- RAI Stages 0 to I. During this early stage of the disease, there is an elevated lymphocyte count (0) and enlarged lymph nodes (I).
- RAI Stage II. In the intermediate stage the spleen becomes enlarged.
- RAI Stages III to IV. In the advanced stage of the disease, bone marrow function is disturbed and there is a significant decrease in red blood cell (III) or platelet counts (IV).
The RAI stages predict survival:
- RAI Stage 0 or I. More than 10 years
- RAI Stage II. Six years
- RAI III or IV. One to two years
New biologic markers, including chromosome abnormalities, can also help with prognosis. Having normal chromosomes, an extra chromosome 12, or a missing part of chromosome 13 are favorable; a missing part of either chromosome 11 or 17 is unfavorable.
In 5 percent to 10 percent of cases, CLL can transform to an aggressive lymphoma. This is called Richter's syndrome, and it requires lymphoma treatment.
Treatments
Most patients with early-stage chronic lymphocytic leukemia do not need any treatment when the disease is first diagnosed. These patients will live 10 to 15 years on average, and early treatment offers no advantage.
Treatment should start when the patient either has an advanced form of the disease or has an intermediate stage with significant symptoms, very enlarged lymph nodes or a rapid increase in the lymphocyte count (doubling in less than 12 months).
Chemotherapy
The most common treatment for CLL is the chemotherapy drug fludarabine. This is given intravenously (through an IV) five days a week once a month, for four to six months. Although the schedule for fludarabine is rather inconvenient, it causes only modest side effects, such as fatigue. Most patients will have a good response to this treatment and remain in remission without further treatment for two to three years.
The remission rate and length of remission are increased by adding a monoclonal antibody, Rituximab – also used to treat lymphoma – to fludarabine. The most aggressive regimen, fludarabine plus cyclophosphamide plus Rituximab (FCR), has the highest response rate (close to 100 percent), but at the expense of a high infection risk. A new chemotherapy drug, bendamustine, was recently approved for CLL; the patient receives two doses intravenously every month. Another agent, lenalidomide (Revlimid), which is FDA-approved for the treatment of multiple myeloma and certain types of myelodysplastic syndrome, has activity in CLL and is an oral medication.
Chlorambucil, an oral chemotherapy drug, may be used instead of fludarabine, especially for elderly or frail patients.
Stem cell transplantation
Allogeneic stem cell transplantation, also called bone and marrow transplantation (BMT), can be used to potentially cure CLL. However, this therapy is only used to treat the occasional young patient with aggressive CLL, since most patients with CLL live so long that the risk of transplant can seldom be justified.
Investigational therapies
UCSF is dedicated to improving outcomes for CLL patients through the use of investigational therapies and clinical research trials. Clinical trials currently available to UCSF patients include Flavopiridol for CLL cases that don't respond to standard treatment. Flavopiridol kills CLL so quickly that blood salt and acid-base abnormalities arise, potentially causing kidney failure. The first cycle of Flavopiridol is given in the intensive care unit with kidney hemodialysis immediately available. Also available are national trials adding biologic agents, such as lenalidomide, to standard accepted CLL therapies.
Learn more about clinical trials and how they work.
Awards & recognition

Best in California and No. 7 in the nation for cancer care
Recommended reading
More treatment info
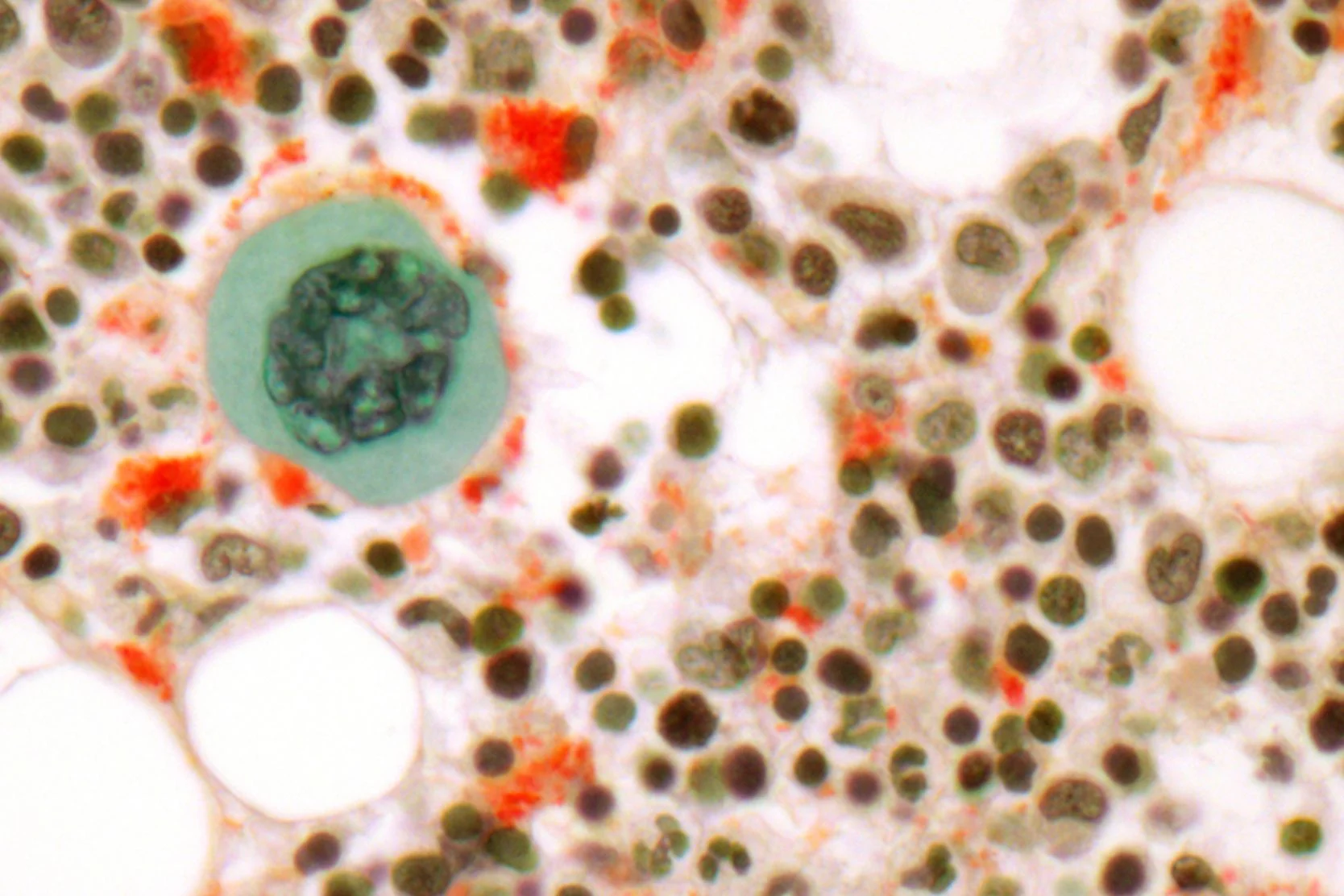
Allogeneic Transplant
Improvements in supportive care over the past 10 years have improved the results and tolerability of blood and marrow transplants (BMT).Learn more here.
Related services & treatments
Specialties
Treatments
- Cancer Chemotherapy
- Allogeneic Stem Cell Transplant
- Clinical Trials
Helpful resources
Support services
UCSF Health medical specialists have reviewed this information. It is for educational purposes only and is not intended to replace the advice of your doctor or other health care provider. We encourage you to discuss any questions or concerns you may have with your provider.